Challenges and Opportunities in Oncolytic Virus Development
Author:
Dr Babaj Yadav
Principal Consultant
Introduction
Oncolytic viruses (OVs) have gained significant attention and interest in the last two decades for their unique ability to selectively infect and replicate within cancer cells. The concept of oncolytic virotherapy involves using viruses to target and destroy cancer cells while sparing normal, healthy cells. OVs not only directly destroy tumor cells but also stimulate the host’s anti-tumor immune system response, which destroy neighbouring cancer cells.
The historical roots of oncolytic viruses in cancer treatment can be traced back to early 1900 observations where a leukemia patient experienced temporary remission of cancer symptoms after contracting a presumed influenza infection. (Dock G., 1904).
Several viruses, including adenoviruses, measles viruses, herpes simplex viruses, Newcastle disease viruses, and vaccinia viruses, have been clinically tested as oncolytic agents. The features of commonly used oncolytic viruses are listed in Table 1. (Lovat and Parker, 2023 and Laucer and Beil 2022).
The emergence of genetic engineering techniques allows for precise modifications or deletions of viral genes, serving a dual purpose in oncolytic virus development. These modifications enhance the virus’s replication competence and target specificity while simultaneously attenuating its virulence and increasing immunogenicity.
Table 1 - Commonly Used Oncolytic Viruses & Their Features
Ds, double stranded; ss, single stranded; kb, kilobase; N/A, not available.
Adapted from Lovat and Parker, 2023 and Laucer and Beil 2022.
To date, various regulatory agencies globally have approved four oncolytic viruses (OVs) and one non-oncolytic virus for the treatment of diverse cancers.
Rigvir, a pioneering OV, secured its first approval for melanoma therapy in Latvia in 2004 (Alberts et al., 2018). Another oncolytic virus, H101, utilizing adenovirus, gained approval for treating nasopharyngeal carcinoma in China in 2005 (Liang et al., 2018). In 2015, the United States Food and Drug Administration (FDA) granted approval for T-VEC (BLA# 125518), designating it as the sole FDA-approved OV for melanoma therapy (Pol et al., 2016).
Recent regulatory milestones include the approval of G47Δ in Japan in 2021 for glioma treatment, and in December 2022, Nadofaragene firadenovec received clearance in the United States for managing bladder cancer.
Table 2 - Approved Oncolytic Viruses Worldwide
Reference: Yang Li et al 2023
Anticancer Mechanism of Ovs
Oncolytic viruses demonstrate diverse mechanisms of anticancer activity, with their effectiveness influenced by intricate interactions among the oncolytic virus, the tumor microenvironment, inflammation status, and the host immune system. The therapeutic efficacy of oncolytic viruses is primarily determined by a combination of direct cancer cell lysis and the indirect activation of anti-tumour host immune responses. Oncolytic viruses employ diverse mechanisms for eliminating tumor cells, involving apoptosis, necrosis, pyroptosis, and autophagy (Li et al., 2023, Lauer and Beil., 2022).
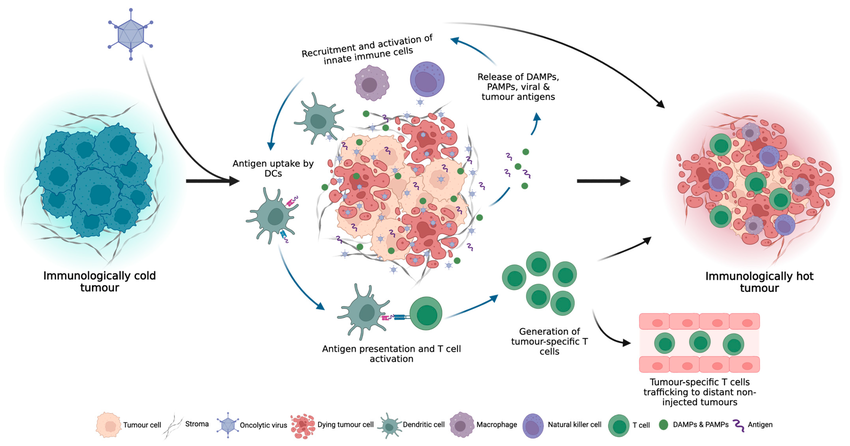
Firstly, oncolytic viruses selectively target and replicate within cancer cells leading to direct lysis of the tumor cells, a phenomenon known as virus-induced direct oncolysis (see Figure 1).
Following the induction of tumor cell death by oncolytic viruses, tumor-specific antigens, damage-associated molecular patterns (DAMPs), and e pathogen-associated molecular patterns (PAMPs) are released. These released components trigger immune host responses, activate anti-tumor immunity, and recruit activated immune cells into the tumor microenvironment. Overall, the release of tumor antigens leads to a systemic anti-tumor immune response throughout the body.
Local inflammation induced by oncolytic viruses also causes destruction within the tumor microenvironment, enabling indirectly mediated cell death of both virus-infected and uninfected cancer cells.
Oncolytic viruses target tumor vasculature-associated endothelial cells, reducing angiogenesis and inducing cytotoxicity.
Replication and amplification of oncolytic viruses within the tumor are major determinants of tumor eradication and lead to the infection of new tumor cells.
Figure 1
Re Mechanisms employed by oncolytic viruses. Int. J. Mol. Sci. 2020, 21, 7505
Legal & Regulatory Framework for OV Products
The development of oncolytic virus products is primarily regulated by the ICH and EMA guideline, “ICH Considerations for Oncolytic Viruses, 2009,” which outlines the scope and extent of Chemistry, Manufacturing, and Control (CMC), nonclinical, and clinical testing. Additionally, FDA guidance on the analysis of OV shedding (FDA, 2015) is pertinent.
For genetically modified OVs, specific guidelines pertaining to gene therapy products apply. For instance, FDA guidelines such as “Preclinical Assessment of Investigational Cellular and Gene Therapy Products” (2013), “Guidance for Industry: CMC Information for Human Gene Therapy Investigational New Drug Applications (INDs)” (2020), and “Considerations for Design of Early-Phase Clinical Trials of Cellular and Gene Therapy Products” (2015) are relevant. Similarly, the EMA’s “Guideline on the quality, non-clinical, and clinical aspects of gene therapy medicinal products” (2018) is applicable.
The general scientific principles outlined in these guidelines are also relevant to the development and testing of OVs that are not genetically modified. Moreover, FDA guidance on clinical considerations for therapeutic cancer vaccines (FDA, 2011) is considered relevant.
It’s essential for developers to adhere to these regulations to ensure the safety, efficacy, and quality of OV products throughout their lifecycle.
Current Clinical Trial Landscape of Oncolytic Viruses:
The clinical landscape of OVs has been extensively reviewed by Lauer & Beil, 2022. An updated analysis of clinical trials documented on clinicaltrials.gov revealed that there are currently over 180 OVs in clinical trials.
By February 2024, there were a total of 188 OV-related clinical trials registered in ClinicalTrials.gov.
The majority of the clinical trials were phase I (n = 99; 52.7 %). There were an additional 12 studies listed as early Phase 1, 34 (18.1%) studies reported as phase I/II, 35 (18.6%) as phase II, 4 (2.1%) as phase III, only 1 (0.5%) as phase II/III and 2 as Phase IV clinical trials. The TOP distribution of indications and countries are listed in Fig. XXX.
There are 44 trials listed for adenovirus, 42 for HSV, 28 for vaccinia virus, 10 for reovirus, 10 for MV, four each for coxsackie, Maraba virus and VSV, and one each for poliovirus, Seneca Valley virus, and parvovirus.
An analysis of OV trials (phase I, II, III, and IV) by indication revealed that the most common indication being trialled was skin cancer (melanoma), with 35 trials documented. This was followed by brain cancer (glioma/glioblastoma; 22 trials), breast cancer (20 trials), head and neck carcinoma (16 trials), lung cancer including non-small-cell lung cancer (16 trials), colorectal cancer (14 trials), pancreatic cancer (13 trials), liver cancer (12 trials), and bladder cancer (11 trials). Advanced solid tumors were covered in up to 40 studies in total.
Regarding the geographical distribution of OV clinical trials, about half of the documented locations were in the USA (91 trials), followed by China (50 trials), Spain (16 trials), Canada (14 trials) and the UK (12 trials).
Challenges of Development of Oncolytic Viruses
One of the primary challenges in delivering oncolytic viruses is the requirement for direct injection into the tumor (intratumoral delivery) to achieve direct therapeutic effect. Intratumoral delivery enables precise control of the oncolytic virus concentration at the target site and prevents potential side effects resulting from mistargeting the virus to other organs (Li et al., 2020).
This method is more suitable for superficial tumors like melanoma, but deeper tumors like glioblastoma pose operational difficulties due to their location and therefore requires skilled technicians to administer the treatment. In addition, IT delivery presents the risk of bleeding and undesired metastasis at the lesion site (Lin et al., 2023).
Finding alternative, efficient delivery methods for oncolytic viruses to reach tumor cells without technical difficulties or patient non-compliance is imperative.
Some additional challenges in OV therapy include (Yang Li et al., 2023);
Presence of Pre-existing Immunity: Some patients may have pre-existing immunity to oncolytic viruses, potentially reducing their therapeutic effectiveness.
Tumor Heterogeneity: Tumors exhibit diverse cell types and genetic mutations, making it challenging to achieve uniform viral infiltration.
Varied Administration Methods: Differences in how oncolytic viruses are administered can affect their distribution within the body and subsequent treatment outcomes.
Concerns Regarding Off-target Toxicity: Certain oncolytic viruses may cause unintended toxicity in healthy tissues, limiting their clinical utility.
Biological Safety Considerations: Using oncolytic viruses raises various biological safety concerns during application, which may pose risks of contamination.
Clinical Challenges:
(Shalhout et al., 2023)
Appropriate selection of patients and lesions. For example, immunocom promised patients may not be good candidates because oncolytic virus-mediated anti-tumour immunity could be compromised in these patients.
Unconventional pharmacokinetics associated with intratumoral administration of Ovs.
Lack of validated clinical endpoints and biomarkers.
Challenges in selecting appropriate control arms for randomized studies due to a lack of approved standard-of-care intratumoural agents.
Careful design of clinical trials to account for differences in pharmacokinetics and potential pseudoprogression.
Regulatory Challenges:
(Shalhout et al., 2023)
Regulatory considerations for OVs encoding transgenes which could potentially qualifying as gene therapy.
Limited preclinical data on non-oncolytic viral gene therapy and differences between OVs, making standardized regulatory guidelines challenging.
Setting regulatory standards for product manufacturing and testing, including quality control procedures.
Requirements for establishing virus origin, derivation history, culture conditions, and safety assessments in early phase trials.
Mandated adherence to aseptic and good manufacturing practices due to biological material.
Safety Profile of OVs in Clinical Development
OVs have been reported to have a tolerable safety profile in clinical trials. The most common treatment related adverse event reported was low grade (CTCAE grade 1–2) fever, chills and rigors, nausea and vomiting, flu- like symptoms, fatigue, and local injection site pain (Macedo et al. 2020).
The most adverse events were comparable across IT and intravenous routes of delivery. Immune- related adverse events (irAEs) were rare and while there were a few reported in the studies, high grade irAEs were uniformly associated with coadministration of immune checkpoint inhibitors (Macedo et al. 2020).
Exploring Clinical Efficacy and Addressing Challenges in OV Therapy
OVs as a single therapy vs combination
Typically, monotherapy using oncolytic viruses (OVs) studied to date, demonstrates some therapeutic effectiveness against cancers with greater immunogenicity, yet it often fails to meet anticipated outcomes. This discrepancy can be attributed, in part, to the fact that most reported OVs either lack transgenes or carry only a single transgene. Consequently, these viruses have limited ability to induce antitumor immune responses. However, the next generation of OV products, which incorporate multiple immune-stimulating transgenes, show potential for enhancing antitumor efficacy (Lin et al., 2023).
Preclinical and emerging clinical data support combination strategies, especially the combination of OVs with immune checkpoint inhibitors (ICIs) (Macedo et al., 2020). Combining OVs with ICIs has garnered interest in recent years due to their potential to offer improved therapeutic outcomes over single agents. OVs also have an acceptable safety profile and a mechanism of action that largely does not overlap with other therapeutic modalities.
A large number of clinical trials on the combination of OVs with various immunotherapies, including ICIs, are currently underway worldwide, showing encouraging overall performance.
OVs eliminate tumor cells through mechanisms distinct from those of other anticancer therapies, making them a rational candidate for combination with most other treatment modalities, including systemic chemotherapies, immunotherapies, targeted therapies, and radiotherapy. Furthermore, the toxicity profiles of OVs are limited and generally do not overlap with those associated with other therapeutic approaches (Shalhout et al., 2023).
The core idea is always to transform the “cold tumor” into the “hot tumor”. Plus, the combination of OV and immune checkpoint blockade, especially PD-1/PD-L1 inhibition, is one of the most frequently adopted approaches and most promising to enter clinical trials that may benefit more patients with “immune desert” tumors.
Figure 2
Lovatt and Parker 2023
Combination of OVs with ICI
The combination of oncolytic viruses (OV) and immunotherapy is currently the most concerning combination strategy.
Oncolytic viruses have the potential to reverse immunosuppression and transform “cold” tumors into “hot” tumors, enhancing the response to ICI treatment. A meta-analysis investigating the combination of oncolytic viruses and ICIs revealed that this approach exhibited improved efficacy and demonstrated enhanced safety when used in conjunction with pembrolizumab (Li et al, 2023).
Patients treated with neoadjuvant oncolytic adenovirus, HSV and vaccinia virus followed by ICIs targeting PD-1, PD-L1 and CTLA-4 have all demonstrated clinical benefit, with durable response rates observed in subsets of patients (Li et al., 2022, Nassiri et al., 2023, Chesney et al., 2023).
In a Phase Ib clinical trial of clinically approved, T-VEC combined with ipilimumab in the treatment of advanced melanoma, the combination therapy was more effective than T-VEC or ipilimumab monotherapy, with an ORR of 50% (Puzanov et al., 2016). In the updated results of the follow-up phase II clinical trial, a significantly improved objective response rate (ORR) was observed with T-VEC-ipilimumab versus ipilimumab with 39% of patients (38/98) in the combination arm and 18% of patients (18/100) in the ipilimumab arm had an objective response (P=0.02) (Chesney et al., 2018). At the 5-year follow-up, T-VEC-ipilimumab continued to provide durable and improved ORR versus ipilimumab in patients with advanced melanoma without additional toxicity. Although, the combination did not provide statistically significant PFS or OS benefits in the overall patient population, this was the first randomized controlled study of the combination of an oncolytic virus and a checkpoint inhibitor that met its primary end point (Chensey et al., 2023b).
T-VEC was also tested in combination with pembrolizumab in treatment-naïve patients with advanced melanoma (MASTERKEY-265) (Chesney et al., 2023a and Long at al., 2016). While phase Ib reports showed promising tumor responses, in the randomized phase III portion, T-VEC-pembrolizumab treatment did not significantly improve PFS (HR 0.86; 95% CI 0.71 to 1.04; p=0.13) or OS (HR 0.96; 95% CI 0.76 to 1.22; p=0.74) compared with placebo-pembrolizumab, although the subgroup-specific PFS trends were similar to those observed in the current study (Chesney et al., 2023a). The authors argued that different checkpoint inhibitors likely have different mechanisms of action, which might explain why T-VEC-ipilimumab conferred significant benefits whereas T-VEC-pembrolizumab did not (Chesney et al., 2023b). The negative results underscore the importance to carefully consider key factors when selecting the combination of oncolytic viruses (OVs) and immune checkpoint inhibitors (ICIs). These factors include the tumor subtype and stage of progression, the criteria for assessing changes in tumor size, the optimal timing for administering OVs and ICIs, and other relevant considerations (Tian et al., 2022).
In another phase II clinical trial of T-VEC combined with pembrolizumab for locally advanced or metastatic sarcoma, the combined therapy showed positive efficacy, with an overall ORR of 35% (Kelly et al., 2020).
Interestingly, clinical studies showed that combination of OV and ICIs lead to an increase in CD8+ and CD4+ TILs and increases in circulating CD8+ and CD4+ T cells, suggesting the presence of a systemic anti-tumour immune response (Fakih et al., 2019, Lillie et al., 2020, Krige et al., 2021, Shoushtari et al., 2023, Ribas et al., 2017, Andtbacka et al., 2018, Li et al., 2022, Nassiri et al., 2023).
Currently, a large number of clinical trials on the combination of OV and various immunotherapies are being carried out around the world, but most of the results are mainly reported in conferences, and the overall performance is encouraging. Among various combination strategies, the prospect of OV combined with immunotherapy is the most promising. (Lin et al. 2023).
Conclusion & Future Direction
In recent years, the field of oncolytic virotherapy has undergone significant advancements, primarily fueled by the distinctive capacity of oncolytic viruses (OVs) to selectively target and replicate within cancer cells. This unique characteristic, coupled with the ability to induce direct tumor cell lysis and stimulate anti-tumor immune responses, positions OVs as promising candidates for novel therapeutic interventions across a spectrum of cancer types. These advancements are particularly crucial for cancers that exhibit resistance to conventional and targeted therapies.
An intriguing aspect of OVs lies in their potential to convert “cold” tumors, typically unresponsive to immune checkpoint inhibitors (ICIs), into “hot” tumors. Furthermore, OVs can serve as versatile platforms for genetic engineering, allowing for the incorporation of immunomodulatory therapeutic genes. This genetic manipulation enhances the capacity of OVs to augment T-cell function against tumors, presenting an innovative approach in cancer therapy.
Rooted in historical observations dating back to the early 1900s, the development of OVs as cancer treatments has been significantly advanced by modern genetic engineering techniques, allowing for precise modifications to enhance their therapeutic efficacy. The regulatory approval of several OVs globally marks substantial milestones in the field, underlining their growing importance in cancer therapy.
However, the clinical landscape of OVs continues to evolve, with ongoing trials investigating their efficacy across diverse cancer types and treatment settings. Challenges in OV therapy include the necessity for direct intratumoral injection, patient-specific immune responses, tumor heterogeneity, varied administration methods, and concerns regarding off-target toxicity and biological safety.
Addressing these challenges demands a comprehensive approach, encompassing advancements in delivery methods, patient selection criteria, and regulatory standards. Collaborative efforts between researchers, clinicians, and regulatory agencies are indispensable for surmounting these obstacles and optimizing the therapeutic potential of OVs.
Oncolytic viruses have demonstrated moderate clinical efficacy in various trials as a single agent, showcasing their potential as standalone treatments for cancer. Moving forward, future directions in OV research should prioritize refining combination strategies, particularly with immune checkpoint inhibitors, which have demonstrated the ability to enhance the anti-tumor immune response. Ongoing clinical trials investigating novel neoadjuvant OV and ICI combinations aim to achieve durable clinical responses, especially in patients who may not derive significant benefit from systemic ICI treatment alone. Additionally, pre-clinical studies utilizing OVs engineered to encode ICI antibodies have yielded promising results, with tumor growth control and overall survival comparable to traditional OV and systemic ICI therapy. While awaiting clinical validation, this targeted and localized expression of ICI antibodies holds promise in overcoming adverse events associated with systemic immunosuppression, a significant limitation of ICI therapy.
As research in this field continues to evolve, further advancements in OV-based therapies are anticipated, paving the way for more effective and safer treatments for cancer patients.
Continued exploration of OV mechanisms of action, optimization of clinical trial designs, and development of standardized regulatory guidelines will further propel the field towards realizing its full potential in cancer therapy.
In conclusion, while significant progress has been achieved in the field of oncolytic virotherapy, challenges persist. With ongoing innovation and collaboration, OVs represent a transformative approach to cancer treatment, offering renewed hope for patients worldwide.
To discuss your Challenges and Opportunities in Oncolytic Virus Development, speak to a member of the team today…